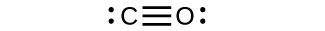
If the second element is oxygen, the trailing vowel is usually omitted from the end of a polysyllabic prefix but not a monosyllabic one (that is, we would say “monoxide” rather than “monooxide” and “trioxide” rather than “troxide”). Normally, no prefix is added to the first element’s name if there is only one atom of the first element in a molecule. Table 4.1 "Numerical Prefixes for Naming Binary Covalent Compounds" lists these numerical prefixes. A system of numerical prefixes is used to specify the number of atoms in a molecule. The second element is named by taking the stem of the element name and adding the suffix - ide. The first element in the formula is simply listed using the name of the element. Naming binary (two-element) covalent compounds is similar to naming simple ionic compounds. For example, we have already seen CH 4, the molecular formula for methane. It is a greenhouse gas, but it is a minor component of Earth’s atmosphere, formed in combustion of carbon-containing materials, in fermentation, in respiration of animals, and employed by plants in the photosynthesis of carbohydrates. Numerical subscripts are used if there is more than one of a particular atom. Lewis dot structures are one way to represent how atoms form covalent bonds. It takes two electrons to make a covalent bond, one from each bonding atom. The configuration of the compound is linear and centrosymmetric. Each of the oxygen atoms is attached to the central carbon atom by a double covalent bond. Covalent bonding is the type of bond that holds together the atoms within a polyatomic ion. Carbon dioxide is a chemical compound made up of a carbon atom and two oxygen atoms. 
Then the other nonmetal symbols are listed. Covalent bonding generally happens between nonmetals. Typically, a molecular formula begins with the nonmetal that is closest to the lower left corner of the periodic table, except that hydrogen is almost never written first (H 2O is the prominent exception). because these compounds exist as separate, discrete molecules. carbon dioxide PCI3 phosphorus trichloride carbon monoxide nitrogen dioxide NO2 hydrogen chloride Nomenclature of Covalent Compounds (using table 3) 1. The chemical formulas for covalent compounds are referred to as molecular formulas A chemical formula for a covalent compound. zip file containing this book to use offline, simply click here. You can browse or download additional books there. More information is available on this project's attribution page.įor more information on the source of this book, or why it is available for free, please see the project's home page. 
Additionally, per the publisher's request, their name has been removed in some passages. However, the publisher has asked for the customary Creative Commons attribution to the original publisher, authors, title, and book URI to be removed. Normally, the author and publisher would be credited here. This content was accessible as of December 29, 2012, and it was downloaded then by Andy Schmitz in an effort to preserve the availability of this book. See the license for more details, but that basically means you can share this book as long as you credit the author (but see below), don't make money from it, and do make it available to everyone else under the same terms. For example, phosphorus exists as P4 tetrahedra-regular polyhedra with four triangular sides-with a phosphorus atom at each vertex.This book is licensed under a Creative Commons by-nc-sa 3.0 license. (b) A few elements naturally exist as polyatomic molecules, which contain more than two atoms. (a) Several elements naturally exist as diatomic molecules, in which two atoms (E) are joined by one or more covalent bonds to form a molecule with the general formula E2. \): Elements That Exist as Covalent Molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
